Answer:
(a) In the single crystal lattice,0.001 % of silicon atoms per unit volume that displaced.
(b)In the single crystal lattice,
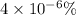 of silicon atoms per unit volume that displaced.
of silicon atoms per unit volume that displaced.
Step-by-step explanation:
Unit cell:
- 8 atoms at the corners at
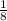 each cell
each cell - 6 atoms in the face at
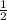 each in cell.
each in cell. - 4 atoms within cell.
Total number of atoms per unit cell of silica is
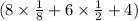 =8 .
=8 .
Dimension of unit cell is
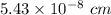 .
.
The volume occupied by a single Si atom in the crystal structure is
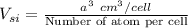
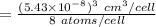
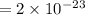
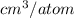
The concentration of Si atom is
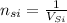
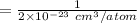
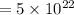
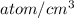
(a)
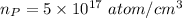
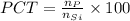
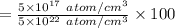
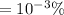
=0.001 %
In the single crystal lattice,0.001 % of silicon atoms per unit volume that displaced.
(b)
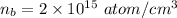
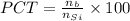
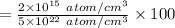
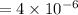 %
%
In the single crystal lattice,
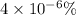 of silicon atoms per unit volume that displaced.
of silicon atoms per unit volume that displaced.