Answer:
Mass of SO₂ can be made from 25.0 g of Na₂SO₃ and 22 g of HCl = 12.672 g
Step-by-step explanation:
SO₂( sulfur dioxide) can be produced in the lab. by the reaction of hydrochloric acid & sulphite salt such as sodium.
the balanced chemical equation is as follows
Na₂SO₃ + 2 HCl → 2 NaCl + SO₂ + H₂O
Moles of Na₂SO₃ =
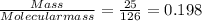
Moles of HCl =
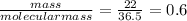
using mole ratio method to find limiting reagent
For sodium sulfite
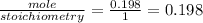
for HCl
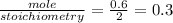
since sodium sulfite is limiting reactant for above chemical reaction
1 mole of Na₂SO₃ produce 1 mole of SO₂
0.198 mole of Na₂SO₃ produce 0.198 mole of SO₂
∴ Mass of SO₂ produce = mole x molar mass of SO₂
= 0.198 x 64
= 12.672 g