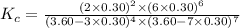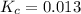Answer: The value of the equilibrium constant, Kc, for the reaction is 0.013
Step-by-step explanation:
Initial concentration of
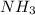 = 3.60 M
= 3.60 M
Initial concentration of
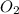 = 3.60 M
= 3.60 M
The given balanced equilibrium reaction is,
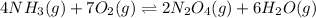
Initial conc. 3.60 M 3.60 M 0 M 0 M
At eqm. conc. (3.60-4x) M (3.60-7x) M (2x) M (6x) M
The expression for equilibrium constant for this reaction will be,
![K_c=([N_2O_4]^2* [H_2O]^6)/([NH_3]^4*[Cl_2])](https://img.qammunity.org/2021/formulas/chemistry/college/40ghbm3x1j111n5dtppj7ltf36qdtjnz9y.png)
![[N_2O_4]=0.60M](https://img.qammunity.org/2021/formulas/chemistry/college/k6nvyf9egdg9sls9yinoq90wo5vfvlye1k.png)
2x = 0.60 M
x= 0.30 M
Now put all the given values in this expression, we get :