Answer:
1.5 litre of C5H12 produce 9 litre of water vapour;
Step-by-step explanation:
First balance the combustion reaction;
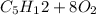 →
→
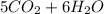
from the balance reacion it is clearly that,
one mole of C5H12 reacts completely with 8 mole of O2 gas;
Hence
one litre of C5H12 reacts completely with 8 litre of O2 gas;
there fore 1.5 litre C5H12 needs 12 litre oxygen gas but we have 15 litre.
so,
C5H12 is a limiing reactant and O2 is a excess reactant.
so quantity of H2O depends on limiting reactant;
one litre of C5H12 produce 6 litre of water vapour;
therefore,
1.5 litre of C5H12 produce 9 litre of water vapour;