Answer:
0.22L
Step-by-step explanation:
Given parameters:
Concentration of solution = 4.00M
Mass of lithium Bromide = 75g
Unknown:
Volume of water = ?
Solution:
Concentration in terms of the molarity is the number of moles of solute in a solution.
Molarity =
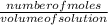
let us find the number of moles first;
Number of moles =
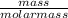
Molar mass of LiBr = 7 + 80 = 78g/mol
number of moles =
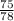
number of moles = 0.96moles
Volume of water =
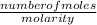 = 0.96/4 = 0.22L
= 0.96/4 = 0.22L