Answer:
The enthalpy change during the reaction is -199. kJ/mol.
Step-by-step explanation:
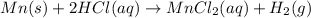
Mass of solution = m
Volume of solution = 100.0 mL
Density of solution = d = 1.00 g/mL
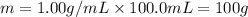
First we have to calculate the heat gained by the solution in coffee-cup calorimeter.
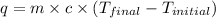
where,
m = mass of solution = 100 g
q = heat gained = ?
c = specific heat =
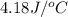
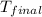 = final temperature =
= final temperature =
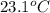
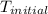 = initial temperature =
= initial temperature =
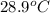
Now put all the given values in the above formula, we get:
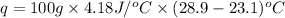
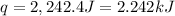
Now we have to calculate the enthalpy change during the reaction.
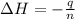
where,
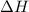 = enthalpy change = ?
= enthalpy change = ?
q = heat gained = 2.242 kJ
n = number of moles fructose =
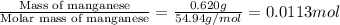
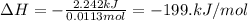
Therefore, the enthalpy change during the reaction is -199. kJ/mol.