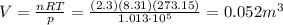Answer:
Step-by-step explanation:
We can solve this problem by using the ideal gas equation, which states that:
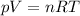
where
p is the gas pressure
V is the volume of the gas
n is the number of moles
R is the gas constant
T is the absolute temperature of the gas
In this problem, we have:
n = 2.3 mol of the gas
The gas is in STP condition, so:
T = 273.15 K is the temperature
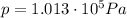 is the pressure
is the pressure
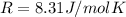 is the gas constant
is the gas constant
Solving for V, we find the volume: