Answer: Concentrations of cyclohexane and methylcyclopentane at equilibrium are 0.0223 M and 0.0027 M respectively
Step-by-step explanation:
Moles of cyclohexane = 0.069 mole
Volume of solution = 2.8 L
Initial concentration of cyclohexane =
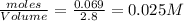
The given balanced equilibrium reaction is,
cyclohexane ⇔ methylcyclopentane
Initial conc. 0.025 M 0
At eqm. conc. (0.025-x)M (x) M
The expression for equilibrium constant for this reaction will be,
K= methylcyclopentane / cyclohexane
Now put all the given values in this expression, we get :
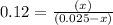
By solving the term 'x', we get :
x = 0.0027
Concentration of cyclohexane at equilibrium = (0.025-x ) M = (0.025-0.0027) M = 0.0223 M
Concentration of methylcyclopentane at equilibrium = (x ) M = (0.0027) M