Answer:
Mass flow = 0.0203 slug/s
Step-by-step explanation:
given data
diameter = 8 in
velocity = 16 ft/s
nitrogen temperature = 90°F = 549.67 °R
gage pressure = 10 psi
atmospheric pressure = 14.7 psi
solution
we know absolute pressure that is express as
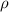 absolute =
absolute =
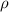 gage +
gage +
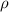 atmospheric ....................1
atmospheric ....................1
put here value we get
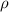 absolute = 10 + 14.7
absolute = 10 + 14.7
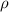 absolute = 24.7 psi
absolute = 24.7 psi
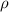 absolute = 3556.8 psf
absolute = 3556.8 psf
and For nitrogen
R = 1775 ft-lb/slug °R
and we get gas equation that is express as
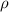 absolute =
absolute =
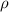 R T ...............2
R T ...............2
3556.8 =
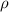 × 1775 × 549.67
× 1775 × 549.67
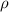 = 3.645 ×
= 3.645 ×
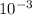 slug/ft³
slug/ft³
so here Mass flow that is express as
Mass flow =
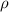 AV .................3
AV .................3
Mass flow = 3.645 ×
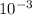 × (
× (
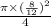 ) × (16)
) × (16)
Mass flow = 0.0203 slug/s