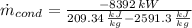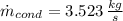Answer:
Step-by-step explanation:
The heat rate received by the cooling water is:
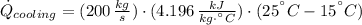
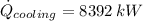
The saturation pressure is 12.532\,kPa at 50 °C. The specific enthalpies for saturated liquid and vapor at given pressure and temperature are, respectively:
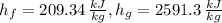
The rate of condensation of the steam in the condenser is: