Answer:
For a: The edge length of the crystal is 533.5 pm
For b: The atomic radius of potassium is 231.01 pm
Step-by-step explanation:
To calculate the lattice parameter or edge length of the crystal, we use the equation:
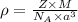
where,
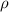 = density =
= density =
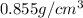
Z = number of atom in unit cell = 2 (BCC)
M = atomic mass of metal = 39.09 g/mol
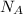 = Avogadro's number =
= Avogadro's number =
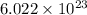
a = edge length of unit cell = ?
Putting values in above equation, we get:
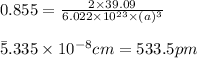
Conversion factor:
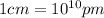
Hence, the edge length of the crystal is 533.5 pm
To calculate the edge length, we use the relation between the radius and edge length for BCC lattice:
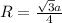
where,
R = radius of the lattice = ?
a = edge length = 533.5 pm
Putting values in above equation, we get:
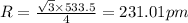
Hence, the atomic radius of potassium is 231.01 pm