Step-by-step explanation:
It is given that 100 kg feed solution contains 30 wt% solution NaOH. Hence, feed contains 70 kg of water and 30 kg of NaOH.
Therefore, mixer outlet shows 5 wt% NaOH.
Let us assume that the mixture outlet be F kg so, by applying mass balance of NaOH we get the value of force as follows.
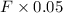 = 30
= 30
F = 600 Kg
In that 30 kg is NaOH and 570 kg is water which also means that initially water present is 70 kg. And, additional water added is 500 kg .
Thus, water feed rate is 500 kg/hr.