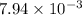Answer : The value of equilibrium constant (Kp) is,
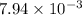
Explanation : Given,
Partial pressure of A at equilibrium = 6.70 atm
Partial pressure of B at equilibrium = 10.1 atm
Partial pressure of C at equilibrium = 3.60 atm
The given chemical reaction is:
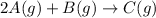
The expression for equilibrium constant is:
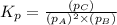
Now put all the given values in this expression, we get:
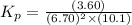
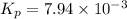
Thus, the value of equilibrium constant (Kp) is,