Answer : The percent yield of the reaction is, 75.6 %
Solution : Given,
Mass of Pb = 451.4 g
Molar mass of Pb = 207 g/mole
Molar mass of PbO = 223 g/mole
First we have to calculate the moles of Pb.
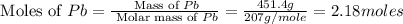
Now we have to calculate the moles of
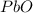
The balanced chemical reaction is,
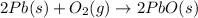
From the reaction, we conclude that
As, 2 mole of
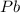 react to give 2 mole of
react to give 2 mole of
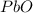
So, 2.18 mole of
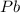 react to give 2.18 mole of
react to give 2.18 mole of
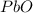
Now we have to calculate the mass of
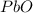
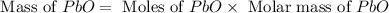
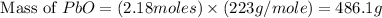
Theoretical yield of
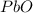 = 486.1 g
= 486.1 g
Experimental yield of
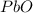 = 367.5 g
= 367.5 g
Now we have to calculate the percent yield of the reaction.
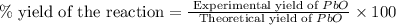
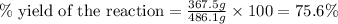
Therefore, the percent yield of the reaction is, 75.6 %