Answer:
Step-by-step explanation:
The number of mole of the hydrogen sample at 27 C and 750 mmHg pressure is:
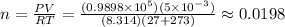 mol
mol
This number of mole will be the same although we change the pressure and temperature. For STP (Temperature = 0 C = 273 K, and Pressure = 760 mmHg = 76 cmHg = 1 Bar = 1.01 x
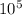 Pa):
Pa):
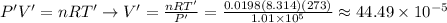 in
in
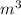 . But in L, we find that V' = 0.4449 L
. But in L, we find that V' = 0.4449 L