Answer:
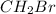
Step-by-step explanation:
Dipole-dipole interaction is a type of intermolecular force that exits in polar molecules. Electronegativity of atoms bonded in polar molecules are different. Because of difference in electronegativity partially charged develops which attracted towards each other through electrostatic force. This electrostatic force between partially charged ions are called dipole-dipole interactions. Dipole moment is a vector quantity.
For, example, HCl
In
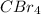 , electronegativities of carbon (C) and bromine (Br) are different and four dipoles exist in the molecules but the net vector sum of all the dipoles are zero. Therefore, its is a non-polar molecule.
, electronegativities of carbon (C) and bromine (Br) are different and four dipoles exist in the molecules but the net vector sum of all the dipoles are zero. Therefore, its is a non-polar molecule.
In the molecule,
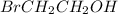 , the strongest intermolecular force is hydrogen bonding.
, the strongest intermolecular force is hydrogen bonding.
In
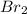 , same atoms are bonded, therefore, it is a non-polar molecule and hence, cannot has dipole-dipole interaction.
, same atoms are bonded, therefore, it is a non-polar molecule and hence, cannot has dipole-dipole interaction.
Therefore, among the given, the only molecule that has dipole-dipole interaction as the strongest intermolecular force is
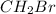 .
.