Answer: The boiling point of solution is 100.53
Step-by-step explanation:
We are given:
8.00 wt % of CsCl
This means that 8.00 grams of CsCl is present in 100 grams of solution
Mass of solvent = (100 - 8) g = 92 grams
The equation used to calculate elevation in boiling point follows:
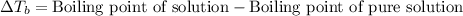
To calculate the elevation in boiling point, we use the equation:
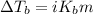
Or,
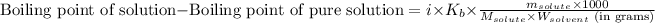
where,
Boiling point of pure solution = 100°C
i = Vant hoff factor = 2 (For CsCl)
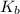 = molal boiling point elevation constant = 0.51°C/m
= molal boiling point elevation constant = 0.51°C/m
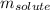 = Given mass of solute (CsCl) = 8.00 g
= Given mass of solute (CsCl) = 8.00 g
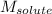 = Molar mass of solute (CsCl) = 168.4 g/mol
= Molar mass of solute (CsCl) = 168.4 g/mol
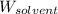 = Mass of solvent (water) = 92 g
= Mass of solvent (water) = 92 g
Putting values in above equation, we get:
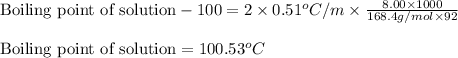
Hence, the boiling point of solution is 100.53