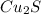Answer: The empirical formula for the given compound is
Step-by-step explanation:
We are given:
Mass of pure compound containing copper and sulfur = 4.963 g
Mass of S = 1.000 g
Mass of Cu = (4.963 - 1.000) g = 3.963 g
To formulate the empirical formula, we need to follow some steps:
- Step 1: Converting the given masses into moles.
Moles of Copper =
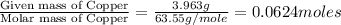
Moles of Sulfur =
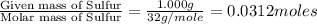
- Step 2: Calculating the mole ratio of the given elements.
For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0312 moles.
For Copper =
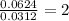
For Sulfur =
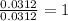
Step 3: Taking the mole ratio as their subscripts.
The ratio of Cu : S = 2 : 1
Hence, the empirical formula for the given compound is