Answer: The percent composition (by mass) of the NaCl (salt) in a binary mixture is 36.8%
Step-by-step explanation:
Binary mixture is a mixture containing two components which are mixed together in any proportion and do not combine chemically with each other.
Mass of mixture=Mass of
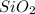 +Mass of
+Mass of
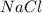 = 23.76 g
= 23.76 g
Mass of
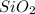 = 15.01 g
= 15.01 g
Mass of
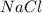 = 23.76 g - 15.01 g =8.75 g
= 23.76 g - 15.01 g =8.75 g
To calculate the mass percentage ,we use the formula:
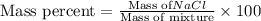
Putting values in above equation, we get:
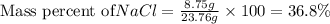
Thus the percent composition (by mass) of the NaCl (salt) in a binary mixture is 36.8%