Answer: The molarity of a solution that is 26.0% by mass in phosphoric acid is 3.06 M
Step-by-step explanation:
Molarity of a solution is defined as the number of moles of solute dissolved per liter of the solution.
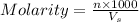
n = moles of solute
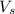 =volume of solution in ml
=volume of solution in ml
Given : 26.0 g of phosphoric acid in 100 g of solution.
To calculate the moles, we use the equation:
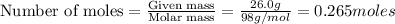
Volume of solution =
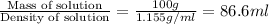
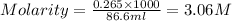
Molarity of a solution that is 26.0% by mass in phosphoric acid is 3.06 M