Step-by-step explanation:
Ionization energy is defined as the energy required to remove the most loosely bound electron from a neutral gaseous atom.
With increase in atomic size of the atom, there will be less force of attraction between the nucleus and the valence electrons of the atom. Hence, with lesser amount of energy the valence electrons can be removed easily.
Since, Na, Mg and Al are all period 3 elements. And, when we move across a period from left to right then there occurs a decrease in atomic size of the atoms. Hence, smaller is the size of an atom more energy is required to remove an electron.
Therefore, out of Na, Mg, the highest
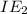 will be that of Na. This is because when sodium will lose one electron then it forms
will be that of Na. This is because when sodium will lose one electron then it forms
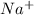 ion which is stable in nature.
ion which is stable in nature.
Hence, in order to remove another electron from
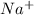 will be difficult. Therefore, it will have high
will be difficult. Therefore, it will have high
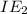 .
.
Similarly, Na will have highest
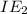 as compared to K and Fe. Also because sodium is smaller in size than K.
as compared to K and Fe. Also because sodium is smaller in size than K.
Since, beryllium is smallest in size as compared to Mg and Sc. Hence, Be will have the highest
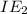 .
.