Answer: The final volume decrease with an increase in pressure at constant temperature
Step-by-step explanation:
According to Boyle's law, pressure of a gas is inversely proportional to the volume of a gas at constant temperature and constant number of moles.
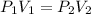
where,
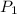 = let initial pressure of gas = p
= let initial pressure of gas = p
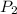 = final pressure of gas = 2p
= final pressure of gas = 2p
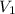 = initial volume of gas = v
= initial volume of gas = v
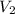 = final volume of gas = ?
= final volume of gas = ?
Now put all the given values in the above equation, we get:
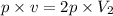
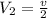
Thus the final volume decrease with an increase in pressure at constant temperature