Answer:
1.05 × 10²⁵ atoms H₂
General Formulas and Concepts:
Atomic Structure
- Reading a Periodic Table
- Moles
Stoichiometry
- Using Dimensional Analysis
Step-by-step explanation:
Step 1: Define
[Given] 35.0 g H₂
[Solve] atoms H₂
Step 2: Identify Conversions
Avogadro's Number - 6.022 × 10²³ atoms, molecules, formula units, etc.
[PT] Molar Mass of H - 1.01 g/mol
Molar Mass of H₂: 2(1.01) = 2.02 g/mol
Step 3: Convert
- [DA] Set up:
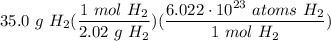
- Simplify [Cancel out units]:
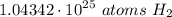
Step 4: Check
Follow sig fig rules and round. We are given 3 sig figs.
1.04342 × 10²⁵ atoms H₂ ≈ 1.04 × 10²⁵ atoms H₂
Topic: AP Chemistry
Unit: Atomic Structure