2.54 moles of CaCl occupies 56.89 liters volume
Step-by-step explanation:
- Each mole of any gas occupies 22.4 liters volume at STP.
- STP is defined as the standard temperature and pressure and those standard values are zero degree and one atmospherics pressure
- 22.4 liters is a constant number as Avogadro's number which is nothing but 6.022*
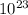 .
. - Hence, to convert 2.54 moles just multiply it with 22.4 L
- That is, 22.4*2.54= 56.89 L
- So, 2.54 moles of CaCl occupies 56.89 L volume space