The question is incomplete, here is the complete question:
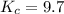 at 900 K for the reaction
at 900 K for the reaction
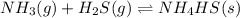
If the initial concentrations of
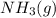 and
and
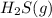 are 2.0 M, what is the equilibrium concentration of
are 2.0 M, what is the equilibrium concentration of
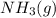 ?
?
Answer: The equilibrium concentration of ammonia is 0.32 M
Step-by-step explanation:
We are given:
Initial concentration of ammonia = 2.0 M
Initial concentration of hydrogen sulfide = 2.0 M
For the given chemical reaction:
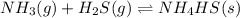
Initial: 2.0 2.0
At eqllm: 2.0-x 2.0-x x
The expression of
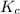 for above equation follows:
for above equation follows:
![K_c=(1)/([NH_3][H_2S])](https://img.qammunity.org/2021/formulas/chemistry/college/i1mr1kc10lo7wbr2j90o3xt02hh2yjwptw.png)
The concentration of pure solids and pure liquids are taken as 1
We are given:
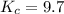
Putting values in above equation, we get:
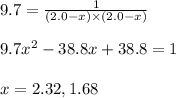
Neglecting the value of x = 1.68 because equilibrium concentration cannot be greater than initial concentration
So, equilibrium concentration of ammonia = 2 - x = (2 - 1.68) = 0.32 M
Hence, the equilibrium concentration of ammonia is 0.32 M