Answer:
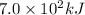 will be released when 0.25 moles of glucose in a can of soda is metabolized
will be released when 0.25 moles of glucose in a can of soda is metabolized
Step-by-step explanation:
Heat of combustion is the amount of heat released on complete combustion of 1 mole of substance.
Given :
Amount of heat released on combustion 1 mole of glucose = 2.8 MJ =
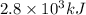
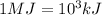
Thus we can say:
1 mole of glucose on combustion releases =
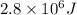
Thus 0.25 moles of glucose on combustion releases =
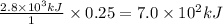
Thus
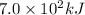 will be released when 0.25 moles of glucose in a can of soda is metabolized
will be released when 0.25 moles of glucose in a can of soda is metabolized