Answer:
The correct answer is option E.
Step-by-step explanation:
Structures for the reactants and products are given in an aimage ;
Number of double bonds in oxygen gas molecule = 1
Number of double bonds in nitro dioxide gas molecule = 1
Number of single bond in in nitro dioxide gas molecule = 1
Number of triple bonds in nitrogen gas molecule = 1
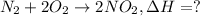
![\Delta H=[2 mol* \Delta H_(f,NO_2)]-[1 mol* \Delta H_(f,N_2)-2 mol* \Delta H_(f,O_2)]](https://img.qammunity.org/2021/formulas/chemistry/college/dp3t107zbl2ef7yhyzgy1grgxluziset7n.png)
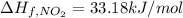
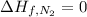 (pure element)
(pure element)
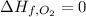 (pure element )
(pure element )
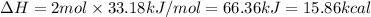
The enthalpy of the given reaction is 15.86 kcal.