Answer:
the solubility of CaCO3 is 0.015g/l 25 °C
is favored at equilibrium
Step-by-step explanation:
The Ksp of calcium carbonate in water at 25 °C is 2.25 x 10-8. CaCO3(s) <----> Ca2+ (aq) + CO3 2- (aq) What is favored at equilibrium?
solubility is the property of a solute to dissolve in a solvent(liquid, gas ) to form a solution(soution can be saturated ,unsaturated, or supersaturated)
CaCO3(s) <----> Ca2+ (aq) + CO3 2- (aq)
in partial dissociation , we can say
2.25x 10^-8=
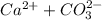
let Ca^2+=CO3^-2=S
2.25x10^-8=S*S
S^2=2.25x10^-8
S=0.00015mol/L
Converting that to g/l
the relative molecular mass of CaCO3=100g/mol
0.00015*100g/mol
0.015g/l
the solubility of CaCO3 is 0.015g/l @room temperature
is favored at equilibrium