Answer: The heat of formation of oleic acid is -94.12 kJ/mol
Step-by-step explanation:
We are given:
Heat of combustion of oleic acid =
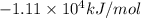
The chemical equation for the combustion of oleic acid follows:
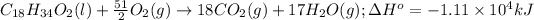
Enthalpy change is defined as the difference in enthalpies of all the product and the reactants each multiplied with their respective number of moles. It is represented as
The equation used to calculate enthalpy change is of a reaction is:
![\Delta H^o_(rxn)=\sum [n* \Delta H^o_f(product)]-\sum [n* \Delta H^o_f(reactant)]](https://img.qammunity.org/2021/formulas/chemistry/college/esjyivbgd1fhj3oo46pyp9cxwjt0l7g52a.png)
The equation for the enthalpy change of the above reaction is:
![\Delta H^o_(rxn)=[(18* \Delta H^o_f_((CO_2(g))))+(17* \Delta H^o_f_((H_2O)))]-[(1* \Delta H^o_f_{(C_(18)H_(34)O_2(l))})+((51)/(2)* \Delta H^o_f_((O_2(g))))]](https://img.qammunity.org/2021/formulas/chemistry/college/xxlgq76eqw8p2ytt97ur3i88s90n4sof30.png)
We are given:
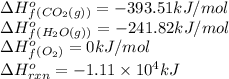
Putting values in above equation, we get:
![-1.11* 10^4=[(18* (-393.51))+(17* (-241.82))]-[(1* \Delta H^o_f_{(C_(18)H_(34)O_2(l))})+((51)/(2)* 0)]\\\\\Delta H^o_f_{(C_(18)H_(34)O_2(l))}=-94.12kJ/mol](https://img.qammunity.org/2021/formulas/chemistry/college/6mxm4tsqhd5sxupradkrj315lg1wginn5o.png)
Hence, the heat of formation of oleic acid is -94.12 kJ/mol