Answer:
B. Maximum velocity of ejected electrons.
Step-by-step explanation:
The ejection of electrons form a metal surface when the metal surface is exposed to a monochromatic electromagnetic wave of sufficiently short wavelength or higher frequency (or equivalently, above a threshold frequency), which leads to the enough energy of the wave to incident and get absorbed to the exposed surface emits electrons. This phenomenon is known as the photoelectric effect or photo-emission.
The minimum amount of energy required by a metal surface to eject an electron from its surface is called work function of metal surface.
The electrons thus emitted are called photo-electrons.
The current produced as a result is called photo electricity.
Energy of photon is given by:
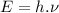
where:
h = Planck's constant
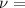 frequency of the incident radiation.
frequency of the incident radiation.