Answer: A volume of 0.592 L of methane gas is required at 237 K and 101.33 kPa when the volume is decreased to 0.50 L, with a temperature of 300 K and a pressure of 151.99 kPa.
Step-by-step explanation:
Given:
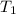 = 237 K,
= 237 K,
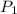 = 101.33 kPa,
= 101.33 kPa,
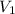 = ?
= ?
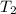 = 300 K,
= 300 K,
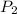 = 151.99 kPa,
= 151.99 kPa,
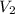 = 0.50 L
= 0.50 L
Formula used is as follows.
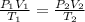
Substitute the values into above formula as follows.
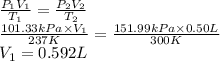
Thus, we can conclude that a volume of 0.592 L of methane gas is required at 237 K and 101.33 kPa when the volume is decreased to 0.50 L, with a temperature of 300 K and a pressure of 151.99 kPa.