Answer: The given chemical reaction can be classified as synthesis and exothermic.
Step-by-step explanation:
A synthesis reaction is defined as the reaction where two small chemical species combine in their elemental state to form a single large chemical species.
Exothermic reactions are defined as the reactions in which heat is released by the reaction. The heat is written on the product side of the reaction.
For the given chemical reaction:
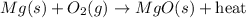
The above chemical reaction is a type of synthesis and exothermic as two substances in their elemental state are combining. Also, heat is getting released in the reaction.
Hence, the given chemical reaction can be classified as synthesis and exothermic.