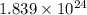Answer: The number of electrons in given amount of silver are
Step-by-step explanation:
The number of moles is defined as the ratio of the mass of a substance to its molar mass.
The equation used is:
......(1)
We are given:
Mass of silver = 7.0 g
Molar mass of silver = 107.87 g/mol
Putting values in equation 1:
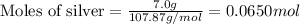
According to the mole concept:
1 mole of a compound contains
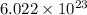 number of particles.
number of particles.
Number of electrons per atom of silver = 47
So, 0.065 moles of silver will contain =
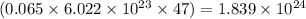 number of electrons
number of electrons
Hence, the number of electrons in given amount of silver are