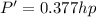Complete Question:
Problem 8 Determine the horsepower required to compress 1 lbm/min of ethylene oxide from 70 °F and 1 atm to 250 psia. The compressor has an efficiency of 75%. The molar heat capacity of ethylene oxide is given by
C_p=10.03+0.0184T C_p[=]Btu/lbmole- "F ; T[=] °F C,
Answer:
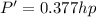
Step-by-step explanation:
From the question we are told that:
Initial Temperature T_1=70 F
Final Temperature
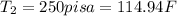
Efficiency
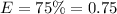
Generally the equation for Work-done is mathematically given by
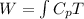
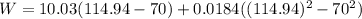
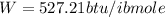
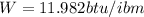
Generally the equation for Efficiency is mathematically given by
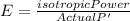
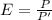
Since
Isotropic Power
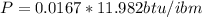
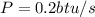
Therefore
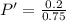
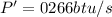
Since
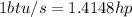
Therefore