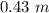Answer:
The molarity will be "0.43 M" and molality will be "0.43 m".
Step-by-step explanation:
Given that:
mass of NaCl,
= 25 g
Volume of solution,
= 1 L
Density of solution,
= 1 gm/cm³
Now,
The weight of solvent will be:
=
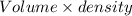
=
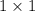
=
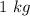
The mole of NaCl will be:
=
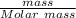
=
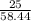
=
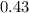
hence,
The molarity will be:
=
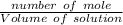
=
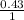
=
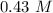
The molality will be:
=
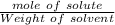
=
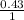
=