It would create 12.1 g of ammonia (NH3).
The amount of hydrogen that is reacted in this reaction is:
(10 g N2)
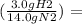 2 g H2
2 g H2
According to the Law of Conservation of Mass, the mass of ammonia produced is the combination of hydrogen and nitrogen reacted or:
2.1g + 10.0g = 12.1 g of NH3