Answer: The molar concentration of 29 g of
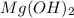 dissolved in 1.00 L of water is 0.497 M.
dissolved in 1.00 L of water is 0.497 M.
Step-by-step explanation:
Given: Mass = 29 g
Volume = 1.00 L
Moles is the mass of substance divided by its molar mass. So, moles of
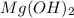 is as follows.
is as follows.
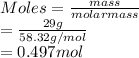
Molarity is the number of moles of a substance divided by volume in liter.
Hence, molarity of the given solution is as follows.
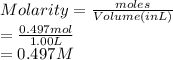
Thus, we can conclude that the molar concentration of 29 g of
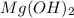 dissolved in 1.00 L of water is 0.497 M.
dissolved in 1.00 L of water is 0.497 M.