Answer:
91150 g
Step-by-step explanation:
First we convert 2.5x10³ g of hydrogen gas into moles, using its molar mass:
- 2.5x10³ g ÷ 2 g/mol = 1250 mol
Then we convert 1250 moles of H₂ into HCl moles, using the stoichiometric coefficients of the reaction:
- 1250 mol H₂ *
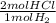 = 2500 mol HCl
= 2500 mol HCl
Finally we convert 2500 moles of HCl into grams, using its molar mass:
- 2500 mol HCl * 36.46 g/mol = 91150 g