Answer:
There are
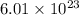 number of molecules.
number of molecules.
Step-by-step explanation:
Given that,
Volume, V = 22.4 L
Temperature, T = 0°C = 273.15 K
Pressure, P = 1 atm = 1.013 bar
We need to find the number of molecules. Let there are n number of molecules. We know that, ideal gas law is as follows :

n = 0.999 moles
No. of molecules,
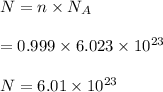
So, there are
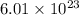 number of molecules.
number of molecules.