Answer: A pressure of 0.681 atm would be exerted by 0.023 grams of oxygen
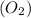 if it occupies 31.6 mL at
if it occupies 31.6 mL at
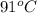 .
.
Step-by-step explanation:
Given : Mass of oxygen = 0.023 g
Volume = 31.6 mL
Convert mL into L as follows.
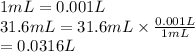
Temperature =
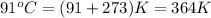
As molar mass of
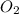 is 32 g/mol. Hence, the number of moles of
is 32 g/mol. Hence, the number of moles of
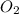 are calculated as follows.
are calculated as follows.
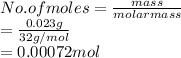
Using the ideal gas equation calculate the pressure exerted by given gas as follows.
PV = nRT
where,
P = pressure
V = volume
n = number of moles
R = gas constant = 0.0821 L atm/mol K
T = temperature
Substitute the value into above formula as follows.
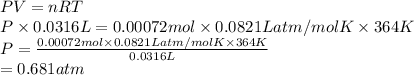
Thus, we can conclude that a pressure of 0.681 atm would be exerted by 0.023 grams of oxygen
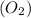 if it occupies 31.6 mL at
if it occupies 31.6 mL at
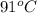 .
.