Answer: The maximum amount of water that can be produced is 0.74 g
Step-by-step explanation:
To calculate the number of moles, we use the equation:
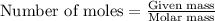 .....(1)
.....(1)
Putting values in equation 1, we get:
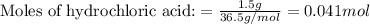
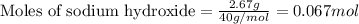
The chemical equation for the reaction is

By Stoichiometry of the reaction:
1 mole of HCl reacts with 1 mole of NaOH
So, 0.041 moles of HCl will react with =
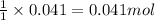 of NaOH
of NaOH
As, given amount of NaOH is more than the required amount. So, it is considered as an excess reagent. Thus, HCl is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
1 mole of HCl produces = 1 mole of water
So, 0.041 moles of HCl will produce =
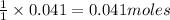 of water
of water
Mass of water=
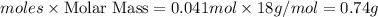
Thus the maximum amount of water that can be produced is 0.74 g