Answer:
The sample's volume at STP is 52.416 L.
Step-by-step explanation:
The STP conditions refer to the standard temperature and pressure. Pressure values at 1 atmosphere and temperature at 0 ° C are used and are reference values for gases. And in these conditions 1 mole of any gas occupies an approximate volume of 22.4 liters.
Then you can apply the following rule of three: if 1 mole of a gaseous sample occupies 22.4 L, 2.34 moles how much volume does it occupy?
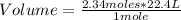
Volume= 52.416 L
The sample's volume at STP is 52.416 L.