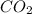Answer: 12.1 ml of ethanol is needed
Step-by-step explanation:
To calculate the moles :
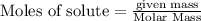
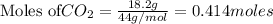
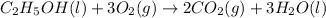
According to stoichiometry :
2 moles of
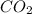 is produced by = 1 mole of
is produced by = 1 mole of
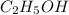
Thus 0.414 moles of
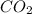 is produced by=
is produced by=
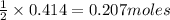 of
of
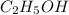
Mass of
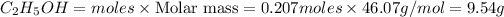
Volume of ethanol =
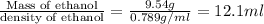
12.1 ml of ethanol is needed to produce 18.2 g of