⇒ Let the width of the the rectangular solid be x since it was not given as a value meaning W=x
⇒It is given that the length is 7 more than the width meaning the length exceeds the width by 7 , L=x+7
⇒The height is 15 decreased by 3 times the size of the width which can be written as H=15 -3(x)
..The formula to calculate the volume of the rectangular solid is given by V=L×W×H
where the Length is (x+7)
where the Width is (x)
where the Height is 15-3x
Volume in terms of width is :
______________________________________________________
V=(x+7)×(x)×(15-3x)
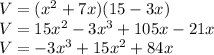
⇒to find the maximum volume you derivate V and equate the derivative to 0 then solve for x
⇒ Derivating we get
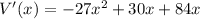
⇒For the maximum volume let V'(x)=0 and solve for x ,Note in this step where you are solving for x you are getting the x-value where V is maximum, to get the volume itself we will plug in the value of x we found in the original equation and simplify to get V
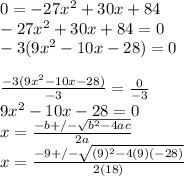
Note since this is a quadratic equation we will have two solutions.
if we simplify further using a calculator we get x= -1.217006237 and 2.328117348
Let us find the maximum volume by plugging the values we got for x .
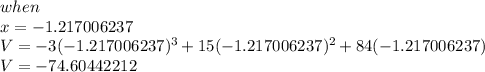
⇒Note this solution cannot be applied since Volume will never be negative.!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!Please keep that in mind
______________________________________________________

⇒The solution is applicable The Maximum volume of the reactangular solid is ≅239.008