The kinetic energy is defined as:
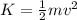
and the potential energy is given by:
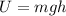
An object will have potential energy if it above ground at certain height and will have kinetic energy if it is moving with some velocity. With this in mind we have:
1.
Since the helicopter is hovering it means that is not moving, then its velocity is zero. Since it is hovering its height is not zero. Therefore, it has GPE but not KE.
2.
Since the bat is hanging it is at a certain height but is not moving, hence it has GPE but not KE.
3.
The statue is at ground level and it is not moving; therefore, it doesn't have either GPE or KE.
4.
Since the ball is kicked along the ground it is moving at certain velocity but it is at ground level, hence it has KE but not GPE
5.
Since the ball is kicked in the air it has both GPE and KE.
6.
Since the car is parked it means is not moving; also it is at ground level therefore, it doesn't have GPE or KE.
7.
Since the cellphone is in your hand by hour head it has certain height; since you are not moving it doesn't have any velocity. Therefore, the cellphone has GPE but not KE.
8.
Since the cellphone is at your head level it has height and since you are walking it is moving, therefore, it has both GPE and KE
9.
The cash register is at ground level and it's not moving, therefore it has both KE and GPE