1) Balance the chemical reaction.
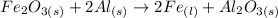
2) List the known and unknown quantities.
Product: Iron (Fe).
Mass: 64.37 g.
Molecular mass: 55.845 g/mol.
Reactant: Aluminum (Al).
Mass: unknown.
Molecular mass: 26.982 g/mol.
3) Convert the mass of Fe to moles of Fe.
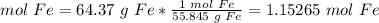
4) Convert moles of Fe to moles of Al
The molar ratio between Fe and Al is 2 mol Fe: 2 mol Al.
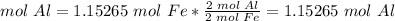
5) Convert moles of Al to mass of Al.
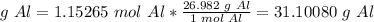
The mass of Aluminum that must be used is 31.10 g Al.
.