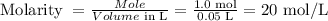Answer
20 mol/L
Step-by-step explanation
Given:
Mass of H₂SO₄ = 98 g
Volume = 0.05 L
From the Periodic Table; Atomic mass (H = 1.0 g, O = 16.0 g, S = 32.0 g)
What to find:
Molarity of the solution.
Step-by-step solution
Step 1: Convert the given mass of 98 g to mole.
The formula to determine the mole is given by:
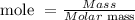
Molar mass of H₂SO₄ = (2 x 1.0 g) + 32.0 g + (4 x 16.0 g) = 98 g/mol
So mole is
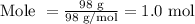
Step 2: Determine the molarity
The molarity formula is given by: