Answer:
Step-by-step explanation:
Given the reaction;
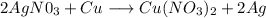
From the reaction, we can see that the Silver (Ag) atom in silver nitrate (AgN0₃) is replaced with copper to produce copper nitrate. In the process, a new element Silver (Ag) is formed.
This type of reaction is known as a single displacement reaction. This type of reaction is the type of reaction in which one element is substituted for another element in a compound, generating a new element and a new compound as products. From the given reaction, we can see that a new compound Cu(NO₃)₂ and an element silver ()