Assuming all 4.75 MnO₂ will react, we have a stoichimetry of 1 to 1 of MnO₂ to MnCl₂, so we would produce the same number of moles of MnCl₂, that is:
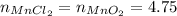
To convert number of moles of MnCl₂ to grams of MnCl₂, we need the molecular weight of it, which can be calculated using the atomic mass of each of its atoms:
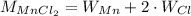
Consulting the atomic masses, we have:
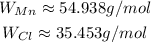
So:
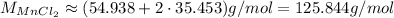
Now, to find the mass, we have:
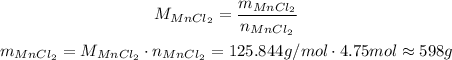
So, approximately 598 grams of MnCl₂ will be produced, assuming complete reaction.