hello
the first step in solving this question is to add the sum of the coupons she presented to the grocery clerk
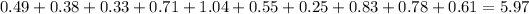
the sum discount on her coupons was $5.97
she bought food worth $39.95
let's subtract the discount from the coupon from the worth of the food
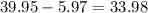
her food bill with coupons would cost her $33.98
the answer to this question is option A